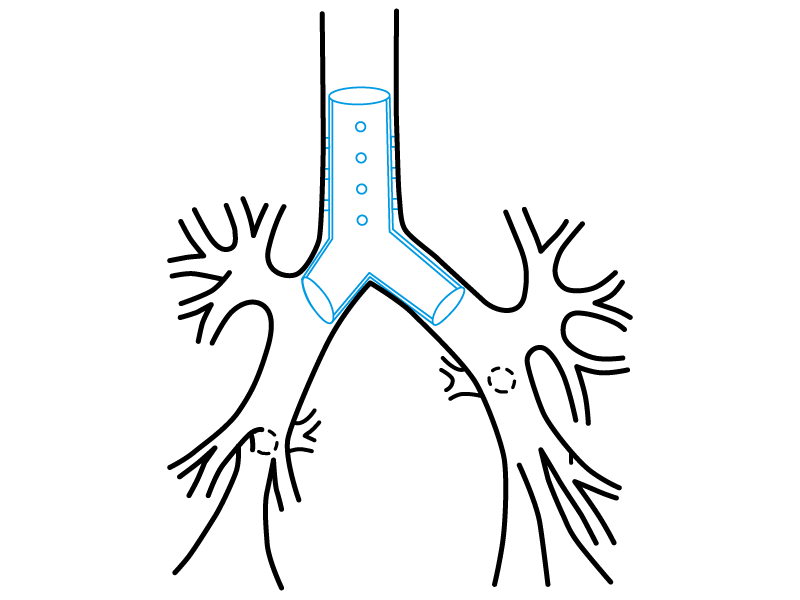
Stening® "Y" Stent
Code SY
Description

Flexible tracheo-carino-bronchial prosthesis for supporting tracheal bifurcation and carinal angle, able to maintain ventilation through the main bronchi in very advanced obstructive conditions.
Indications:
- Tracheal neoplasms.
- Long tracheobronchial neoplasm, with or without a compromise for the carina and/or its slopes.
- Neoplasms that affect both sources.
- Esophageal carcinoma with an airway invasion.
- Tracheoesophageal or tracheocutaneous fistula.
- After laser resection, cryotherapy or electrocautery, to maintain the airway opened.
- Extrinsic compression or compromise of the submucosa.
- Tracheal stenosis.
- Tracheobronchial stenosis.
- Tracheobronchomalacia.
- Amyloidosis.
- Excessive dynamic compression of the airway.
It also admits other uses according to doctor’s judgment due to its length and designing.
The Stening® “Y” has been used with success in tracheotomised patients and in mechanical respiratory assistance, in combination with tracheostomy cannula, to allow ventilation in seriously ill patients when other methods are not possible.

Due to the characteristics of the production process, the
measurements of the devices can vary by +/- 2%
- Medical grade silicone
- Bevelled edges to prevent granulomas
- Spur system to prevent migration
- Removable
- Surface of maximum softness to avoid adherence of secretions
- Transparent or Radiopaque
Implant:
The process will take place under general anaesthesia. This kind of implants must be carried out by experienced staff. The stent can be fitted on a special forceps that is used for “Y” prostheses implant.
Lubricate the forceps’ margin with lidocaine gel. Introduce it inside de stent in order to make its valves penetrate into its bronchial branches (see the photo on previous page). Ventilate the patient with oxygen until you reach the higher saturation possible. After that we will proceed to the patient extubation by removing the tracheoscope from the airway. Immediately, and with the laryngoscope help, the forceps will be guided towards the trachea. When you close the forceps’ valves the bronchial branches from the stent will join, and, in this position, it will pass through the vocal cords towards the trachea. The manoeuvre will continue to displace the forceps-stent assembly inside the trachea until it approaches to the carina. When the edge of the forceps-stent assembly is close to the tracheal carina, the valves must open kindly so that they can notice the prosthesis arrival to the tracheal bifurcation.
At this moment, the valves will open completely so that the stent bronchial branches can be introduced inside the source bronchus. Then the button from the forceps that holds the stent against the carinal edge will be pushed at the same time the forceps is removed.
The manoeuvre must be fast, knowing that it is carried out while the patient is extubated and in apnoea.
To make sure that the stent does not go to the oesophagus and that it cross the vocal cords safely, use direct optic vision.
To verify if the stent passes through the glottis, the optic must accompany the forceps in a parallel way during the manoeuvre.
This is only possible if a second surgeon holds the laryngoscope in a suitable intubation position, whilst the bronchoscopist holds the forceps with his right hand and the optic with his left hand.
Other ways of implant are possible; for instance, the insertion of a bended prosthesis inside a bronchoscope with the right size, then pushing the stent through the bronchoscope interior with a alligator forceps or a smaller diameter bronchoscope when it is closer to the tracheal carina.
The manoeuvre can be completed by adjusting the prosthesis with a forceps.
Removal:
We will proceed to the patient’s intubation with a tracheoscope.
The extraction is easier.
The stent must be taken by its proximal border with a strong forceps to remove it kindly by pulling the forceps and removing the prosthesis at the same time you remove the tracheoscope.
- Despite its size, the Stening® Y stent are well-tolerated. However, its larger length increases the difficulty of getting rid of secretions, and even more when the cough is not effective. Perform nebulisations frequently and if you notice an increase on bronchial secretions attend your kinesiologist daily.
- The appearance of excessive cough may suggest an unwanted touch of only one or both stent bronchial branches with the inflamed bronchial mucosa. If the symptom persists or becomes irrepressible in spite of the anti-inflammatory treatment, removing the stent may be necessary and then introduce a new one with the bronchial branches length shortened.
- Take care of your oral hygiene and treat cavities.
The product should not be reused because this can cause cross contamination.
Polymeric tracheal/bronchial stent, non-bioabsorbable, sterile
A sterile non-bioabsorbable tubular device intended to be implanted into the trachea and/or a bronchus/bronchiole to maintain luminal patency, typically used in cases of obstructions/stenoses, fistulae, tumours, scarring, surgical resection and anastomosis, or pulmonary transplantation. It is made entirely of a synthetic polymer(s) [e.g., silicone] and may have various designs (e.g., semi-soft continuous tube, covered or non-covered mesh structure, straight or branched configuration) intended to conform to the endotracheal/endobronchial surface. It may be expandable in situ (e.g., self-expands) and disposable devices intended to assist implantation may be included.
46977